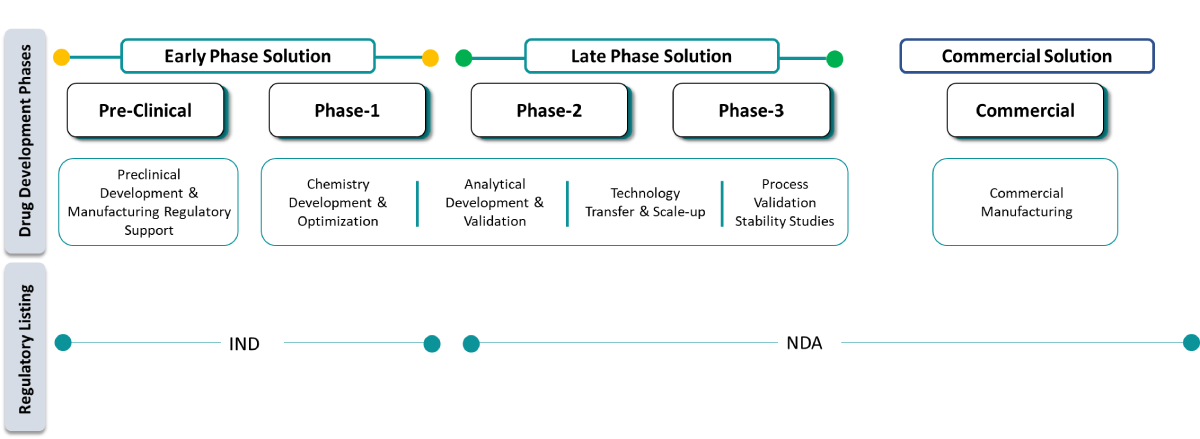
At Shilpa Pharma, we are committed to supporting pharma and biotech companies in developing New Chemical Entities by providing end-to-end solutions across the entire drug development lifecycle while maintaining the highest standards of safety, quality, and efficiency.
Our expertise in route scouting, proof-of-concept studies, process research, risk assessment, and scale-up ensures that we deliver high-quality, scalable, and regulatory-compliant drug substances.
We specialize in identifying and developing the most efficient synthetic routes at the early stage of a program. Our experienced scientists and process engineers focus on evaluating different pathways to optimize yield, purity, and cost-effectiveness.
Through detailed feasibility studies and a phase-appropriate approach, we ensure that the chosen route aligns with the requirements of early-stage research, clinical trials, and commercial manufacturing.

Our POC studies validate the feasibility of the selected synthetic route and ensure smooth scale-up and regulatory acceptance. By conducting small-scale manufacturing, we swiftly supply initial batches for biological testing, stability evaluation, and toxicological studies.
Our team ensures process robustness, enabling smooth extrapolation to higher scales while maintaining regulatory compliance — forming the foundation for process optimization and commercial scalability.

With state-of-the-art facilities and experienced teams, we develop and optimize robust manufacturing processes for NCEs. Our expertise covers route optimization, reaction refinement, and impurity profiling — ensuring scalable, efficient synthesis.
By implementing Quality by Design (QbD) principles and identifying Critical Process Parameters (CPPs) and Critical Material Attributes (CMAs), we ensure batch consistency and regulatory compliance from development to commercialization.

Our scale-up capabilities ensure seamless transition from lab to pilot and full-scale manufacturing. We optimize processes for reproducibility, yield improvement, and cost efficiency, maintaining robustness across scales.
Our GMP facilities enable pilot-scale batch production essential for regulatory submissions and commercial readiness. Through strong process validation and technology transfer, we ensure global regulatory alignment and scalability.

A robust risk assessment strategy — incorporating HAZOP and HAZAN — ensures safety, compliance, and scalability across all NCE development phases. We evaluate potential risks related to materials, reaction conditions, and thermal hazards.
By integrating predictive models and quantitative hazard assessments, we deliver safe, reproducible, and globally compliant processes, minimizing risks and ensuring reliability throughout the product lifecycle.

At Shilpa Pharma, we offer end-to-end contract development and manufacturing solutions, ensuring seamless progression from concept to commercial.
With over 35 years of expertise, state-of-the-art infrastructure, and a commitment to regulatory excellence, we provide high-quality, scalable, and compliant drug substance manufacturing services tailored to the evolving needs of the pharmaceutical industry.




At Shilpa Pharma, we support pharma, mid-pharma, biotech, and academia in accelerating early development through pre-clinical development and manufacturing. Our expertise in route scouting, feasibility studies, and small-scale nGMP & GMP production enables partners to access early-stage material for toxicological and proof-of-concept studies.
Our scientific team optimizes synthetic routes and develops robust processes to ensure a smooth transition from concept to commercialization. Our analytical capabilities ensure method development, validation, and reliability of early-stage data.

Our clinical supply manufacturing services ensure seamless progression of drug candidates across all clinical phases with GMP-compliant drug substances. We provide phase-appropriate manufacturing, ensuring scalability and compliance at every stage.
Through process optimization and flexible manufacturing capabilities, we efficiently produce clinical trial materials aligned with global regulatory standards, ensuring consistent batch production and smooth transitions to late-stage development.

We specialize in manufacturing PV batches that are essential for regulatory submissions and commercial readiness. Our expertise in process optimization, CPP identification, and batch consistency ensures robust and reproducible manufacturing.
Our regulatory-driven approach ensures compliance with USFDA, EMA, PMDA, MFDS, and global health authorities — supporting smooth NDA and ANDA approvals with high-quality, scalable processes.

Our Chemistry, Manufacturing & Controls services play a critical role in enabling successful IND and NDA filings. We deliver comprehensive CMC documentation covering process development, analytical method validation, impurity profiling, and stability studies per ICH guidelines.
Our regulatory experts ensure data integrity and compliance, helping pharma companies streamline the approval process and accelerate their drug development timelines.

At Shilpa Pharma, we provide large-scale manufacturing for KSMs, RSMs, Intermediates, APIs, peptides, and specialty chemicals. Our cGMP facilities meet the highest international standards for quality and regulatory compliance.
With expertise in HPAPI manufacturing and advanced containment technologies, we ensure consistent, compliant, and efficient production for global pharmaceutical partners.

Ensuring a seamless transition from development to commercial manufacturing is central to our tech transfer expertise. We develop strategies that minimize risks while maintaining quality, scalability, and compliance.
Our customized supply security solutions ensure uninterrupted drug availability through strategic inventory management, regulatory compliance, and global supply chain resilience.



Please Note: We partner exclusively with pharmaceutical manufacturers, biotech companies, and international trading firms. Minimum order quantities apply*