Antibody Drug Conjugate Development: How ADCs Are Redefining Targeted Cancer Therapy
Cancer treatment has come a long way, but it still has a fundamental problem.
Conventional chemotherapy does not discriminate. It targets rapidly dividing cells, which means it attacks tumour cells and healthy ones at the same time. The result? Severe side effects, toxicity, and a significant impact on patient quality of life.
For decades, oncologists and drug developers have been searching for a smarter approach: one that delivers a lethal dose of therapy precisely to the cancer cell, while leaving healthy tissue untouched.
That answer is antibody drug conjugate (ADC) development, and it is quickly becoming one of the most exciting frontiers in modern pharmaceutical science.
What Is an Antibody Drug Conjugate (ADC)?
An antibody drug conjugate is often described as a “guided missile” for cancer therapy. It is a type of targeted biologic drug that combines:
- A monoclonal antibody that recognises and binds to a specific antigen on the surface of cancer cells
- A highly potent cytotoxic payload (a chemotherapy drug) that kills the cancer cell once delivered
- A chemical linker that holds the two together during circulation in the bloodstream and releases the payload at the right time
The concept is elegant. The antibody acts as a homing device, seeking out only cells that express the target antigen. Once it binds to the cancer cell, the entire conjugate is internalised, the linker is cleaved, and the toxic payload is released, killing the cell from within.
The result is targeted killing with significantly reduced systemic toxicity compared to traditional chemotherapy.
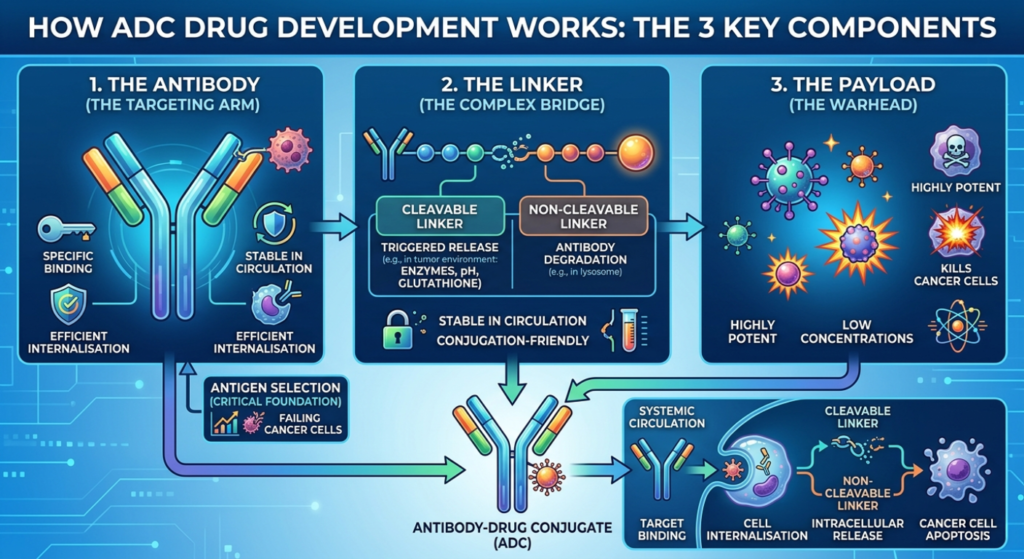
How ADC Drug Development Works: The 3 Key Components
Understanding ADC drug development requires a closer look at each of its three components, because the success of an ADC depends on how well all three work together.
1. The Antibody
The monoclonal antibody forms the targeting arm of the ADC. It must:
- Bind specifically and strongly to an antigen that is overexpressed on tumour cells
- Have minimal cross-reactivity with normal tissues
- Be stable in systemic circulation and capable of efficient internalisation upon binding
Selecting the right antibody is the foundation of the entire programme. Poor antigen selection remains one of the leading reasons why ADC candidates fail in clinical trials.
2. The Linker
The linker is the bridge between the antibody and the payload, and it is arguably the most complex piece of the puzzle.
A good linker must be:
- Stable in circulation: it should not release the payload prematurely while in the bloodstream
- Cleavable in the tumour environment: it must release the drug efficiently once inside the cancer cell
- Conjugation-friendly: it must attach to the antibody without affecting its binding activity
There are two broad types: cleavable linkers (triggered by pH, enzymes, or glutathione in the tumour environment) and non-cleavable linkers (released only after antibody degradation inside the cell).
At Shilpa Pharma, we manufacture a comprehensive range of high-quality ADC linkers, including cleavable and non-cleavable variants, designed to meet the demanding specifications of ADC programmes from discovery to commercial scale.
3. The Payload
The cytotoxic payload is the “warhead” of the ADC. It must be potent enough to kill cancer cells at very low concentrations, since only a small amount of drug reaches the tumour via the antibody.
Common payload classes include:
- Microtubule disruptors (e.g., auristatins like MMAE, MMAF; maytansinoids like DM1, DM4)
- DNA-damaging agents (e.g., calicheamicin, pyrrolobenzodiazepines or PBDs, duocarmycins)
- Topoisomerase I inhibitors (e.g., DXd, used in Enhertu)
Each payload class comes with its own synthesis challenges, potency profile, and toxicity considerations. The drug-to-antibody ratio (DAR), typically between 2 and 8, must be precisely controlled to balance efficacy and safety.
Shilpa Pharma offers a robust catalogue of ADC payloads, including highly potent cytotoxic compounds synthesised under strict containment using HPAPI-grade manufacturing infrastructure.
ADC Therapeutics Pipeline: What's Happening in 2024–25?
The ADC therapeutics pipeline has never been more active. After years of slow progress following early setbacks with first-generation ADCs, the field has exploded, driven by better linker-payload chemistry, improved manufacturing processes, and a deeper understanding of tumour biology.
Approvals and Commercial Success
As of 2025, over 14 ADCs have received regulatory approval globally, including landmark drugs such as:
- Enhertu (trastuzumab deruxtecan): approved for HER2-positive breast cancer, gastric cancer, and now being explored in multiple solid tumours
- Kadcyla (ado-trastuzumab emtansine): one of the first ADCs to demonstrate clear clinical benefit in HER2+ breast cancer
- Trodelvy (sacituzumab govitecan): approved for triple-negative breast cancer and urothelial cancer
- Padcev (enfortumab vedotin): a top-performing ADC in urothelial carcinoma
What the Pipeline Looks Like
There are currently over 150 ADC candidates in active clinical development globally. The pipeline spans a wide range of tumour types: breast cancer, lung cancer, haematologic malignancies, ovarian cancer, and increasingly, non-oncology indications like autoimmune diseases.
Key pipeline trends include:
- Bispecific ADCs: targeting two antigens simultaneously to reduce resistance
- Dual-payload ADCs: delivering two different cytotoxic agents in one construct
- Site-specific conjugation: creating more homogeneous ADCs with better therapeutic windows
- Next-generation linkers: with improved stability and programmable release profiles
The global ADC market, valued at approximately USD 9 billion in 2023, is projected to exceed USD 30 billion by 2030, driven by a wave of new approvals and expanding indications.
For biotech startups and pharma R&D teams, the opportunity window is wide open.
Challenges in Antibody Drug Conjugate Development
Despite the promise, ADC drug development remains one of the most technically demanding disciplines in pharmaceutical science. Understanding the challenges is critical for anyone building an ADC programme.
1. Manufacturing Complexity
ADCs are not small molecules, and they are not standard biologics. They sit at the intersection of both, requiring expertise in:
- Monoclonal antibody production (biologic manufacturing)
- Synthesis of highly potent small molecule payloads (HPAPI-grade chemistry)
- Precision conjugation under controlled conditions
- Rigorous analytical characterisation to verify DAR, aggregation, and purity
This complexity means that specialised CDMO partners with ADC-specific capabilities are essential, as very few organisations can handle the full chain in-house.
2. Linker Instability and Off-Target Toxicity
Premature payload release in circulation is a major cause of ADC toxicity and clinical failure. Getting linker chemistry right, ensuring it is stable in blood but labile in the tumour, is an ongoing area of innovation.
3. Antigen Heterogeneity
Tumours are not uniform. Within a single tumour, not all cancer cells express the target antigen at the same level. This heterogeneity can lead to incomplete tumour killing and resistance over time.
4. Regulatory and Analytical Demands
ADCs are regulated as complex combination products. Regulatory agencies like the USFDA, EMA, and CDSCO require extensive analytical characterisation including:
- Average DAR and DAR distribution
- Aggregation and fragmentation profiles
- Payload and linker identity and purity
- Stability data under multiple conditions
Having robust analytical services in place from the start of development, and not just before filing, is non-negotiable for a successful ADC programme.
How a CDMO Partner Accelerates ADC Drug Development
Not every CDMO is equipped for ADC work. The molecules are complex, the materials are hazardous, and the analytical demands are stringent. Choosing the right CDMO partner from the beginning can make the difference between a programme that advances quickly and one that stalls at the development stage.
A capable ADC CDMO partner should offer:
- Custom synthesis of linkers and payloads: with the ability to handle both early-stage milligram quantities and scale-up to kilograms
- HPAPI-grade manufacturing: with appropriate containment, operator safety protocols, and validated cleaning procedures
- Conjugation expertise: with experience across both stochastic and site-specific conjugation chemistries
- Comprehensive analytical services: including DAR determination, aggregation profiling, forced degradation studies, and stability testing
- Regulatory support: with experience filing and responding to CMC queries for complex biologics and combination products
- IP protection: critical when handling novel payload and linker candidates that are core to a company’s pipeline
Shilpa Pharma Lifesciences offers all of the above through its integrated CDMO services, supported by dedicated analytical capabilities and a proven track record with global pharma and biotech clients.
Why Partner with Shilpa Pharma for ADC Development?
At Shilpa Pharma Lifesciences, we understand that every ADC programme is different, and that the stakes are high. We have built our capabilities specifically to support the complexity of modern ADC drug development, from early discovery to commercial supply.
Here is what sets us apart:
Specialised ADC Components: We manufacture a broad range of linkers and payloads used in ADC programmes, with full characterisation and batch documentation.
HPAPI Manufacturing Excellence: Our HPAPI manufacturing infrastructure is designed to handle highly toxic cytotoxic compounds safely and compliantly at all scales.
Regulatory-Grade Analytical Services: From DAR analysis to stability studies, our analytical team supports every stage of your ADC development timeline.
End-to-End CDMO Partnership: We are not just a supplier. We are a full-spectrum development and manufacturing partner that works alongside your team from molecule to market.
Proven Regulatory Compliance: Zero-observation USFDA audits and a consistent track record with global clients from North America, Europe, and Asia.
India Advantage: Competitive pricing, fast turnaround, and a deep talent pool in synthetic and process chemistry, all from a GMP-certified facility.
Whether you are a biotech startup building your first ADC candidate, an R&D team scaling from discovery to Phase I, or an established pharma company looking for a reliable Indian CDMO for ADC components. We are ready to partner with you.
Conclusion:
Antibody drug conjugate development is no longer an emerging technology. It is a proven, FDA-approved therapeutic modality that is reshaping the oncology treatment landscape and rapidly expanding beyond cancer into autoimmune and inflammatory diseases.
The ADC therapeutics pipeline is richer than ever before. The science is maturing. The manufacturing ecosystem is catching up. And antibody drug conjugate development in India is becoming a serious competitive alternative to traditional Western development centres.
For companies that want to move fast, stay lean, and work with a CDMO that truly understands the complexity of ADC programmes, Shilpa Pharma Lifesciences is your answer.

